
In the highly regulated and high-pressured world of medical device development, getting from concept to final production requires precision, expertise, and compliance with strict regulatory standards. This is where medical device contract manufacturing comes in – a solution that allows companies to bring high-quality medical devices to market without handling every step of the manufacturing process in-house.
As the medical device contract manufacturing market continues to grow, more businesses are partnering with specialist providers to improve efficiency, ensure compliance, and scale production sustainably.
Understanding Medical Device Contract Manufacturing
A medical device contract manufacturer is a company that produces medical devices on behalf of another business under a formal medical device contract manufacturing agreement.
In principle, the medical device company (the Original Equipment Manufacturer (OEM)) provides the design specification and the contract manufacturer handles the manufacturing and production of the device on their behalf. This model – often referred to as contract medical device manufacturing – allows OEMs to retain ownership of their intellectual property while outsourcing production.
For medical devices, this process is highly regulated. Medical device contract manufacturers must comply with standards such as ISO 13485 (the international quality management system for medical devices), FDA regulations for the US market, and UKCA and CE marking requirements for UK and European markets, certifying that a device meets the safety, health, and environmental requirements.
By partnering with trusted medical device contract manufacturers UK or internationally, medical device companies can ensure that their products meet these standards while reducing the costs associated with in-house production.
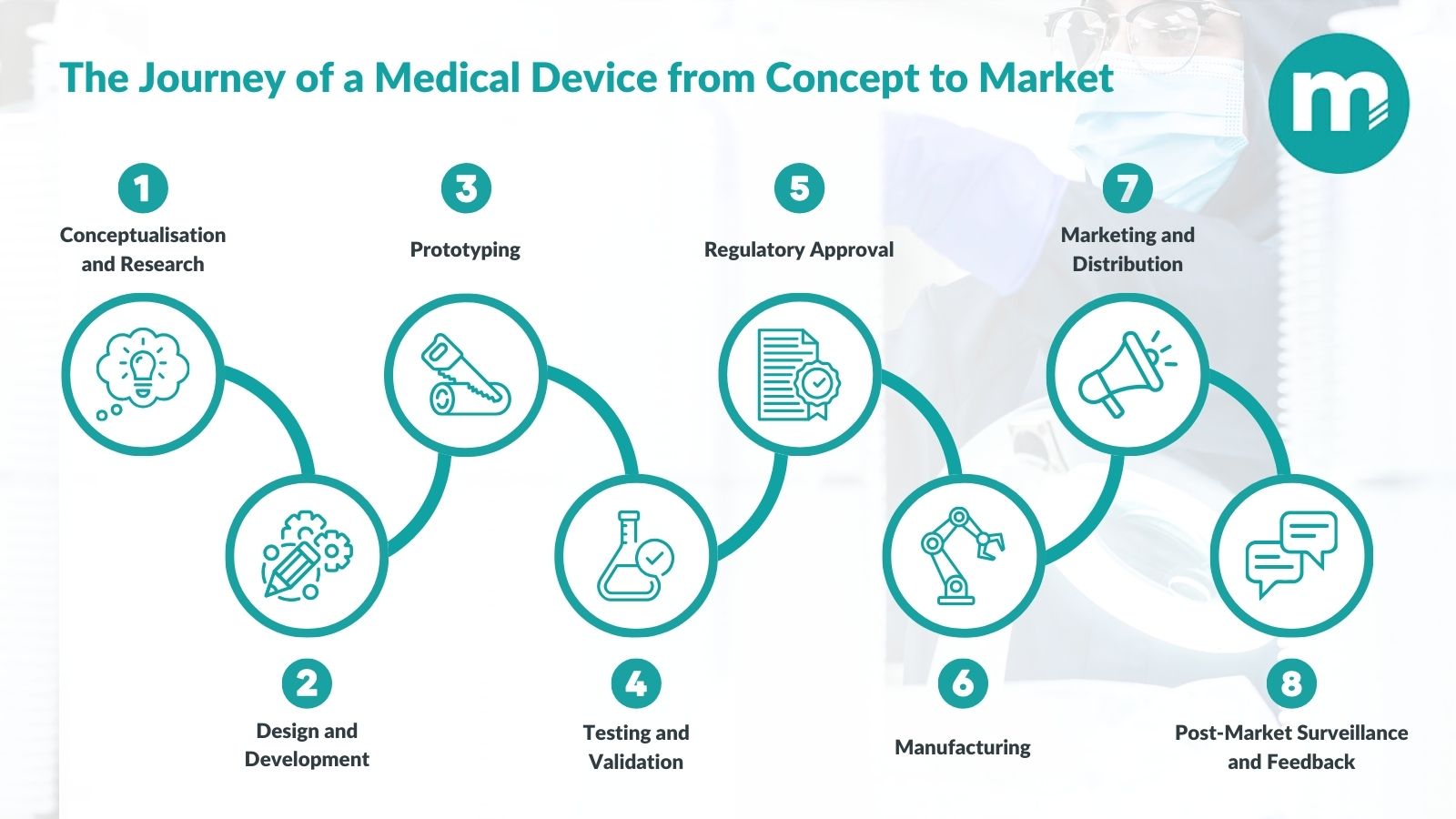
Bringing a medical device to market involves multiple stages and a good medical device contract manufacturing company can guide you through the process from start to finish. Starting with the initial concept, the device is designed and prototyped, with input from the contract manufacturer on materials, manufacturability and any regulatory requirements.
The contract manufacturer validates the production process, manages quality control, and scales up manufacturing to meet demand, ensuring the device is safe, compliant, and ready for market, allowing the client to focus on sales and innovation.
Types of Devices Produced Through Contract Medical Device Manufacturing
Contract manufacturers support a wide range of products across the contract manufacturing medical device industry. Capabilities vary between medical device contract manufacturing companies, depending on facilities and areas of specialism.
For example, providers may support:
- Medical device contract manufacturing Class I products (low-risk, non-invasive devices)
- Contract medical device manufacturing Class II products (moderate-risk devices requiring additional regulatory controls)
- Class III Medical Devices (high risk devices, designed to support and sustain human life)
- Injection moulded medical components
- Cleanroom-assembled devices
- Devices incorporating electronics
- Reusable metal instruments
Understanding your device classification is important when selecting the right contract medical device manufacturers for your project.
Why Companies Choose Medical Device Contract Manufacturers
There are several reasons why medical device companies turn to medical device contract manufacturing services, particularly as the medical device contract manufacturing market size continues to expand globally.
Specialised Expertise
Contract medical device manufacturers often have advanced production capabilities, cleanroom environments, and staff trained specifically in regulated medical device production. This expertise can be difficult and costly to replicate in-house. By outsourcing manufacturing, companies can concentrate on research, design and marketing, leaving production to specialists.
Scalability
Whether producing a limited batch for clinical trials or a high-volume run for commercial launch, medical device contract manufacturing companies provide flexible production capacity.
Cost Efficiency
Outsourcing production eliminates the need for heavy capital investment in machinery, cleanrooms, and compliance infrastructure. While unit costs may appear lower in-house at very high volumes, the overall cost structure – including Overall Equipment Effectiveness (OEE), scrap, regulatory requirements and validation costs – often makes contract medical device manufacturing more economical.
Common Misconceptions About Contract Medical Device Manufacturing
A few misconceptions often arise when companies consider working with medical device contract manufacturers:
- “We lose control over quality.”
In reality, reputable contract medical device manufacturers should deliver the same or better quality than in-house production. The customer sets the quality targets and the manufacturer must operate within validated ISO 13485 systems to meet them.
- “We lose control over our IP.”
Using a reputable company you trust, alongside a properly structured and signed medical device contract manufacturing agreement and NDA, all help to protect your intellectual property.
- “It’s cheaper to produce in-house.”
While unit costs may eventually be lower by manufacturing in-house at scale, the initial capital outlay, equipment validation, regulatory infrastructure, OEE losses, scrap and other operational costs must all be considered. In many cases, medical device contract manufacturing services provide a lower total cost until high production volumes are consistently achieved.
You can learn more about typical medical device manufacturing costs here:
https://www.meridian-medical.com/resources/medical-device-manufacturing-costs/
Choosing the Right Medical Device Contract Manufacturers UK
Selecting the right medical device contract manufacturing company requires careful consideration. When evaluating medical device contract manufacturers UK, look for providers who:
- Have experience in your specific medical device category and classification (Class I or Class II)
- Maintain certifications such as ISO 13485 and FDA registration
- Hold additional accreditations such as ISO 14001 and Cyber Essentials
- Offer transparent communication and collaborative project management
- Can scale production up or down to match your business needs
- Provide engineering, regulatory and production documentation support
Not all medical device contract manufacturing companies offer the same capabilities, so alignment with your product, market, and growth plans is essential.
Medical Device Contract Manufacturing at Meridian Medical
At Meridian Medical, we support medical device manufacturers by handling aspects of production that fall outside their core capabilities. As an experienced UK-based medical device contract manufacturing company, we specialise in injection moulding, cleanroom assembly, validated manufacturing processes and regulatory-compliant production.
Frequently Asked Questions About Medical Device Contract Manufacturing
What is medical device contract manufacturing?
Medical device contract manufacturing is when a specialist third-party company produces medical devices on behalf of another business.
What is the meaning of contract manufacturer?
A contract manufacturer is a company that produces goods for another business under a formal agreement.
What is an example of a contract manufacturer?
An example would be a company that provides injection moulding and cleanroom assembly for a Class II medical device.
What is the difference between a contract manufacturer and a manufacturer?
A manufacturer typically produces products under its own brand, while a contract manufacturer produces goods on behalf of another company.
Speak to a UK Medical Device Contract Manufacturing Expert
If you’re exploring medical device contract manufacturing for your next project, our team can help you evaluate the right production strategy.
Contact us today to discuss how Meridian Medical can support your next medical device project. Get in touch by filling out our online form or contacting us at 01903 732344 or info@meridian-medical.com.



