Medical Pouch Packaging for devices
Medical Pouch Packaging for devices SERVICES
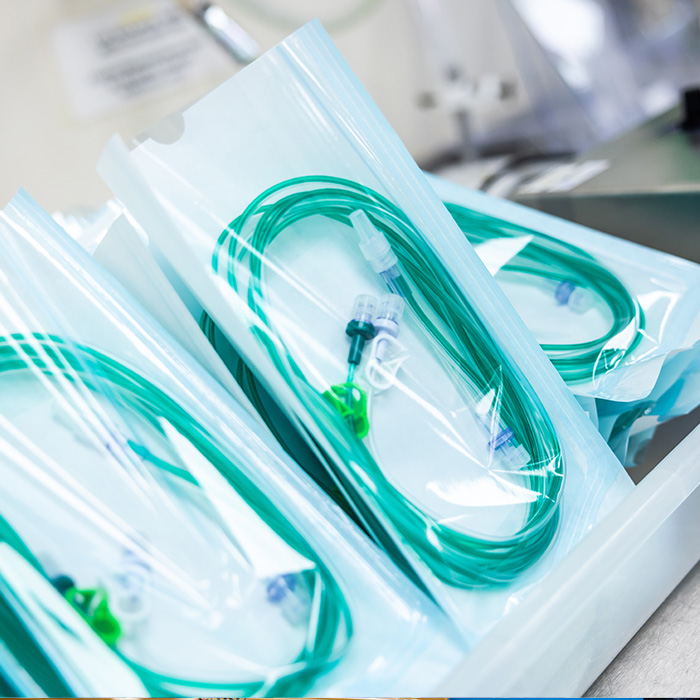
Medical Device Pouch Packaging Services
At Meridian Medical, we provide specialist medical device pouch packaging services using validated, medical-grade packaging materials sourced from our approved suppliers. Whether packaging a single device or assembling a complex kit containing multiple components, our tailored pouch packaging processes are designed to protect product sterility, and maintain product integrity for its shelf life.
All pouch packaging activities are carried out within our class 7 cleanroom environments, which help to ensure that medical devices have low bioburden and are free from foreign particles.
ISO-Compliant Medical Pouch Packaging
Poor medical device packaging can compromise the sterility and performance of even the most precisely engineered products, creating significant risks to patient safety. Exposure to microbial contamination, including pathogenic bacteria and other harmful microorganisms, can be life-threatening, particularly for immunocompromised or critically ill patients.
For this reason, medical device pouch packaging, which is the validated sterile barrier system (SBS), must be carried out in full compliance with ISO 11607 and relevant regulatory standards. Peel pouches and other pouch configurations must be carefully selected, validated and controlled to maintain barrier integrity throughout the product lifecycle.
Cleanroom Medical Pouch Packaging Capabilities
All medical device pouch packaging at Meridian Medical is performed within our ISO Class 7 (Class 10,000) cleanroom environment, ensuring a controlled, contamination-free setting.
We package a diverse range of medical devices into high-performance packaging pouches manufactured from our pre-approved materials, including Tyvek® and cost-effective laminated PE / Paper. Each pouch configuration is selected to ensure compatibility with the intended sterilisation method and to maintain long-term sterility.
Our medical pouch packaging services support both:
- Medical devices manufactured on-site at Meridian Medical
- Customer-supplied devices requiring packaging under controlled cleanroom conditions, and then sterilisation.
All pouches can be supplied pre-printed or labelled with your custom artwork and branding, including GTIN barcodes, QR codes, and lot and expiry data.
How We Ensure Quality and Compliance
Meridian Medical is an ISO 13485-certified manufacturer specialising in medical disposables. We operate under a robust quality management system that governs every stage of the medical pouch packaging process.
Key quality assurance practices include:
- Verification of packaging material integrity by using seal strength peel testing equipment.
- Pre-production seal validation with dye, peel, burst and seal measurements.
- Compliance checks against applicable regulatory standards and customer specifications.
Our dedicated Process and Product validation team oversee all medical packaging services, ensuring every packaged medical device meets required safety, sterility and performance standards before release. This provides confidence that each product is fully compliant and safe for end use.
Case Study: Restoring Reliable Production with Meridian Medical
Discover how Meridian Medical quickly and efficiently resolved the production problems one company was experiencing with an overseas supplier.
Reshoring Medical Product Manufacturing to Overcome Quality Issues
Why Medical Pouch Packaging Is Essential
Medical pouch packaging plays a critical role in protecting medical devices from manufacture through to point-of-use. Properly designed sterile barrier packaging safeguards products against contamination, moisture and physical damage during storage and transportation.
Without effective pouch packaging, microbial contamination can compromise device safety, posing serious risks to patients. High-quality medical device pouch packaging is, therefore, essential to maintaining product integrity and protecting patient health.
Materials Used in Medical Device Packaging Pouches
Medical device pouch packaging utilises validated materials selected for their sterility, durability and barrier performance. Common materials include:
- Tyvek® 1073B, 1059b and 2fs
- Medical-grade papers with Polyethylene and polyester films
- Foil and polymer laminate structures
Our team works closely with customers to select materials that meet sterility requirements, barrier protection needs and regulatory obligations.
Examples of Medical Device Packaging Pouches
We offer a wide range of sterile and non-sterile medical device pouch packaging solutions, including:
- Double pouching for theatre products
- Multiple small packs of pouched products to a single large pouch
- Custom-designed pouch packaging tailored to specific medical applications
Each pouch packaging solution is developed to align with medical device requirements, sterilisation methods and regulatory compliance.
Talk to Us About Your Pouch Packaging Requirements
To find out more about how Meridian Medical can support your custom medical device pouch packaging needs, get in touch with our team today.
You can also contact us by filling out our online enquiry form, calling +44 (0) 1903 732 344, or emailing info@meridian-medical.com.
